solubility
The solvent is something like water. The solute is something the solvent dissolves.
A solution is a homogeneous mixture of at least two substances, where one is dissolved in another.
When water is the solvent, the solution is called an aqueous solution.
In an asymmetrical molecule the more polar functional groups present on a molecule, the more polar the overall molecule is, and therefore, the more soluble the molecule is in polar solvents.
Consider the structures of Vitamin C and Vitamin D2. Whilst both molecules have at least one polar functional group (circled in red), Vitamin C has
5 more polar groups than Vitamin D2, and is therefore significantly more polar than Vitamin D2. In turn, Vitamin C will dissolve in polar solvents like water, whilst Vitamin D2 will dissolve in non–polar solvents such as hexane.
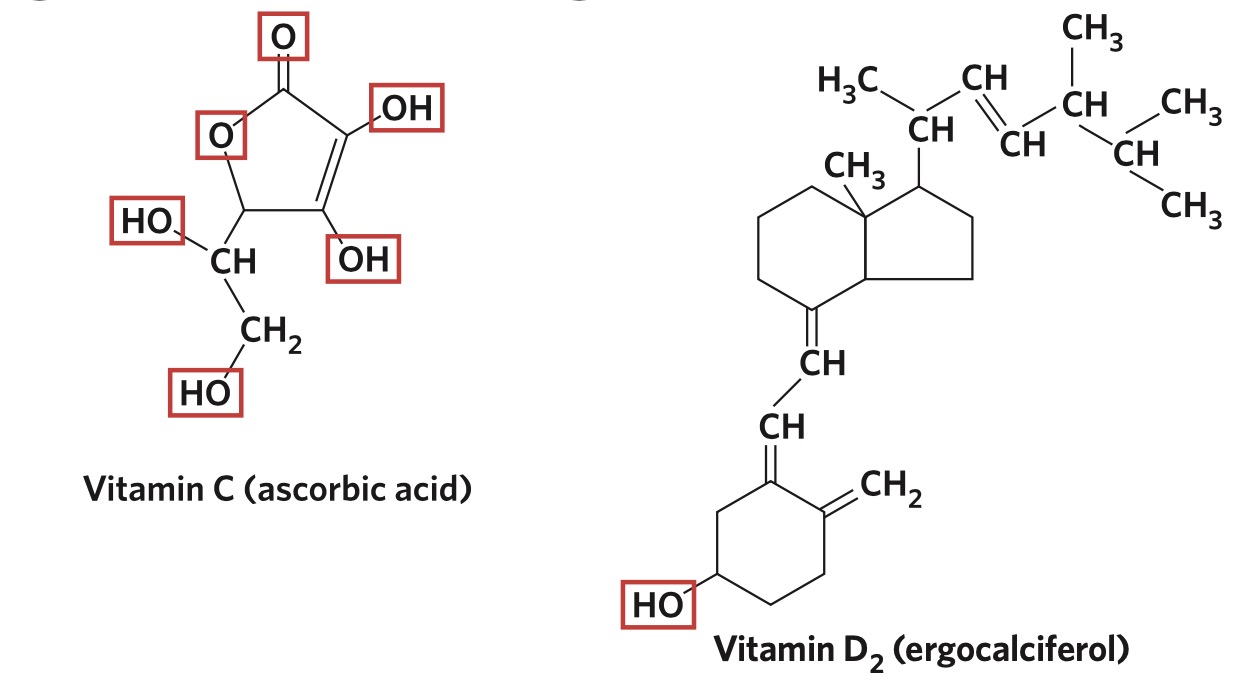
Relatively non-polar molecules such as Vitamin D2 would be insoluble in a polar solvent.
ionisation
While most polar molecules dissolve by dipole-dipole attractions using polarity, some compounds can dissolve using ionisation.
Ionisation is the process in which ions are formed in a reaction. For example, HCl has a very electronegative chlorine atom. When placed in water, oxygen atom in water is attracted to the partially positive hydrogen atom in HCl. Two partially positive hydrogen atoms in water are attracted to the electronegative chlorine atom in HCl.
Therefore, the HCl will separate into H+ and Cl- ions. They are saturated which means the water molecules will surround them.